We'll divide chemistry into 3 sections and 13 chapters:
UNIT 1 (Chap. 1-4)
1. Materials
2. Elements, Compounds and Mixtures
3. Solutions
4. Separating Mixtures
UNIT 2 (Chap. 5-8)
5. Acids and Bases
6. The Air and Oxygen
7. Carbon dioxide
8. Water
UNIT 3 (Chap. 9-13)
9. Atomic Structure
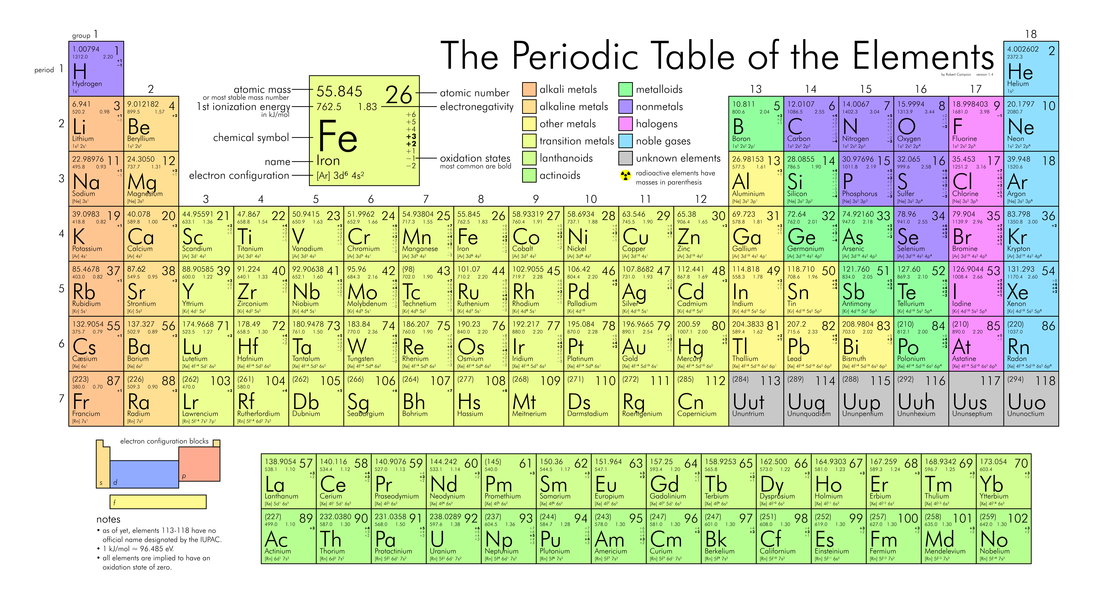
10. The Periodic Table
11. Chemical Bonding
12. Chemical Formulas and Chemical Equations
13. Metals and Plastics
UNIT 1 (Chap. 1-4)
1. Materials
2. Elements, Compounds and Mixtures
3. Solutions
4. Separating Mixtures
UNIT 2 (Chap. 5-8)
5. Acids and Bases
6. The Air and Oxygen
7. Carbon dioxide
8. Water
UNIT 3 (Chap. 9-13)
9. Atomic Structure
10. The Periodic Table
11. Chemical Bonding
12. Chemical Formulas and Chemical Equations
13. Metals and Plastics
Unit 1
20. Materials
21. Elements, Compounds and Mixtures
22. Solutions
23. Separating Mixtures
20. Materials
21. Elements, Compounds and Mixtures
22. Solutions
23. Separating Mixtures
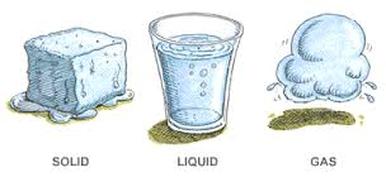
Chapter 1: Materials
- The materials that make anything up is called matter.
- Matter is anything that occupies space and has mass.
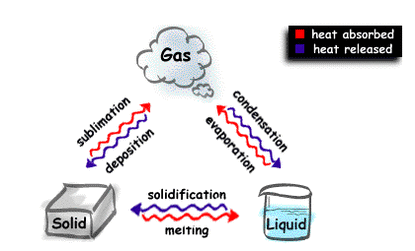
- There are 3 states of matter: Solids, Liquids and Gases
Chapter 2: Elements, Compounds and Mixtures
- Elements: Can't Be split by chemical means (Like letters of the alphabet)
- Compounds: Made up of 2 or more elements (Like letters make a word)
- Mixtures: Made up of 2 or more compounds (Like words make a sentence)
|
Elements:
All the elements are on the Periodic Table of Elements. Elements make up everything around us Eg*
|
Compounds:
Compounds are made of 2 or more elements Chemically Combined. Eg*
|
Mixtures:
Mixtures are made of 2 or more compounds mixed together but NOT chemically combined. Eg*
|
Experiments:
|
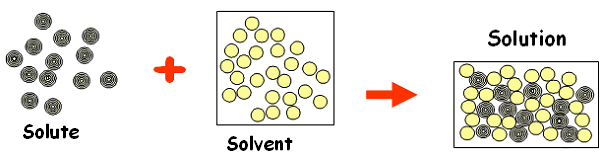
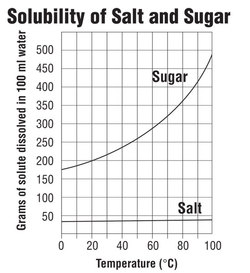
Chapter 3: Solutions
|
Chapter 4: Separating Mixtures
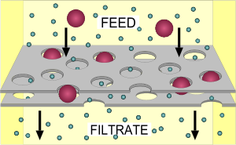
Here we need to learn how to obtain a Pure substance from a mixture. (ie. an element or compound)
There are many ways of separating Mixtures:
Here we need to learn how to obtain a Pure substance from a mixture. (ie. an element or compound)
There are many ways of separating Mixtures:
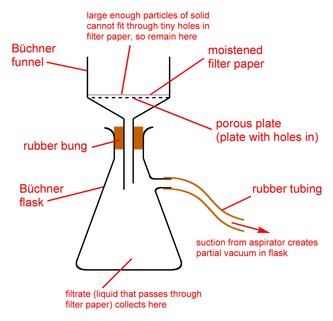
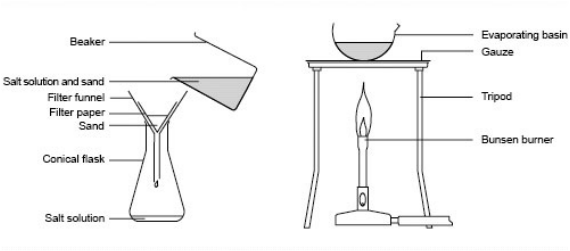
- Filtration
- Evaporation
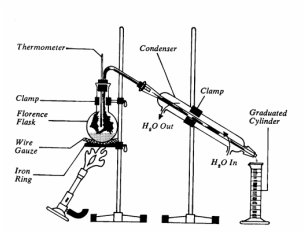
- Distillation
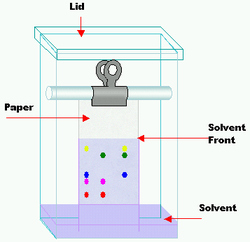
- Paper Chromatography
2. Evaporation
Evaporation is the changing of a liquid into a gas.
Evaporation is the changing of a liquid into a gas.
- Obtaining sea salt from sea water.
UNIT 2
24. Acids and Bases
25. The Air and Oxygen
26. Carbon Dioxide
27. Water
24. Acids and Bases
25. The Air and Oxygen
26. Carbon Dioxide
27. Water
Chapter 5:Acids and bases
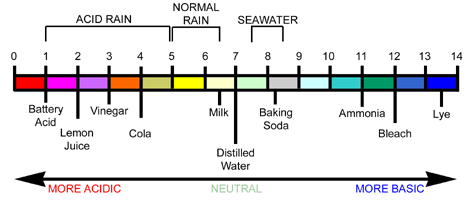
Substances are classified as either acidic or basic
- (Substances that are neither acidic nor basic are classified as being neutral)
- Acidic substances have a pH less than 7
- Basic substances have a pH over 7
- Neutrals have a pH of 7
The pH scale
- The pH scale is used to indicate the level of acidity or basicity of a substance
Indicators
- Indicators are substances that change colour depending on whether they are in an acidic or basic solution
- E.G Litmus paper, universal indicator
- Litmus paper goes from red to BLUE in a BASE and from blue to RED in an ACID
- Universal indicators are a mixture of indicators which will change to a variety of colours depending on the PH substances being tested, tells you exactly how acidic a substance is
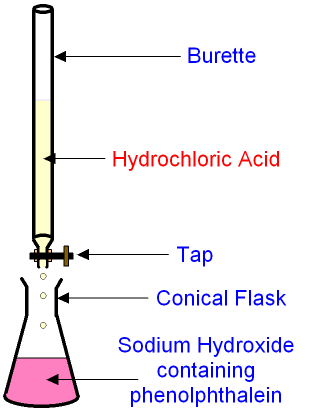
- Phenolphthalein is also an indicator but you DO NOT have to know this for the junior cert
Properties of acids and bases--->
Acids -
Bases -
Reactions of Bases and Acids -
Acid + A metal -------> Salt + Hydrogen
(2HCL+ZN--------------> ZnCL2 + H20 = example of a reaction between acid (hydrochloric acid) and metal (zinc)
NOTE hydrogen is always produced when a metal reacts with an acid the test for hydrogen is that it burns with a pop.
To see hydrogen "popping" https://www.youtube.com/watch?v=ihF_8zNZT74
Acid + Base ---------> Salt + Water
HCL + NaOH -------->NaCL + H20 = Reaction between hydrochloric acid and sodium hydroxide
Acid + Carbonate -------> Salt + Water + Carbon dioxide
2HCL + CaCO3 -----------> CaCL2 + H20 + CO2 = Reaction between hydrochloric acid and calcium carbonate
Neutralisation-
e.g HCL + NaOH ---------------->NaCL +H20
Examples of neutralisation are: - Indigestion tablets
- Adding lime to soil
-Rubbing a dock leaf on a nettle sting
Acids -
- Sharp feel
- pH <7
- Some are corrosive
- React with bases to form salt and water
- Sour taste
- Every day acids include lemons, rain water, vinegar
- Lab examples include Hydrochloric acid (HCL), Sulfuric acid (H2S04) and Nitric Acid (HNO3)
Bases -
- pH >7
- Soapy feel
- Some are corrosive
- Everyday examples include Toothpaste and Bleach and Oven cleaner
- Lab examples include Sodium Hydroxide (NAOH) and Limewater (CA(OH)2)
Reactions of Bases and Acids -
Acid + A metal -------> Salt + Hydrogen
(2HCL+ZN--------------> ZnCL2 + H20 = example of a reaction between acid (hydrochloric acid) and metal (zinc)
NOTE hydrogen is always produced when a metal reacts with an acid the test for hydrogen is that it burns with a pop.
To see hydrogen "popping" https://www.youtube.com/watch?v=ihF_8zNZT74
Acid + Base ---------> Salt + Water
HCL + NaOH -------->NaCL + H20 = Reaction between hydrochloric acid and sodium hydroxide
Acid + Carbonate -------> Salt + Water + Carbon dioxide
2HCL + CaCO3 -----------> CaCL2 + H20 + CO2 = Reaction between hydrochloric acid and calcium carbonate
Neutralisation-
- When an acid and a base react with each other they become neutralised i.e they have a PH of 7
e.g HCL + NaOH ---------------->NaCL +H20
Examples of neutralisation are: - Indigestion tablets
- Adding lime to soil
-Rubbing a dock leaf on a nettle sting
A Titration ------> Is an accurate way of neutralisation
Chapter 6-The air and oxygen
Air is a mixture of gasesL
- 78% Nitrogen
- 21% oxygen
- 1% carbon dioxide , water vapour and other gases
Oxygen and Carbon dioxide
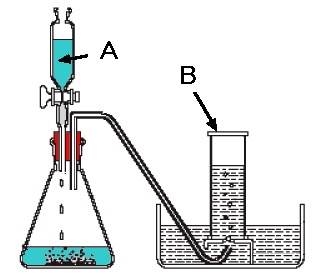
- Oxygen can be prepared in the lab using Manganese dioxide as a Catalyst (something which changes the speed of a chemical reaction without getting used up itself)
The reaction can be summarised in the following chemical equation
2H202----MnO2-------->O2 +2H20
Properties of Oxygen
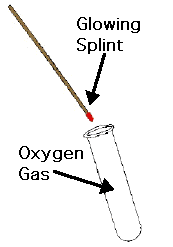
Test for oxygen gas = Oxygen will relight a glowing splint
2H202----MnO2-------->O2 +2H20
Properties of Oxygen
- PHYSICAL PROPERTIES
- Colourless, odourless, tasteless
- Slightly soluble in water
- Slightly more dense than air
- CHEMICAL PROPERTIES
- Neutral gas
- Supports combustion
- Reacts with metals to form basic oxides - e.g MgO
- Reacts with non metals to form acidic oxides - e.g C02
Test for oxygen gas = Oxygen will relight a glowing splint
Uses of oxygen -
% of oxygen in air:
- Approx. 1/5 of air
- Needed for aerobic respiration (see biology section)
- Needed for burning fuels
- Used in welding metals
% of oxygen in air:
- Approx. 1/5 of air
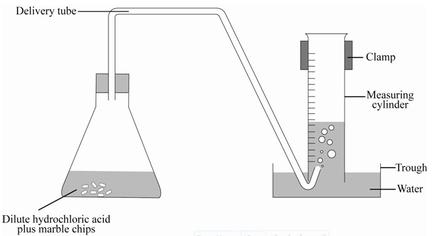
Chapter 7-Carbon dioxide
Lab preparation of carbon dioxide (CO2)
Word equation = CaCO3 + 2 HCL ---------> CaCL2 + H2O +CO2
Properties of carbon dioxide
Physical
The test for carbon dioxide is that it turns limewater milky
Properties of carbon dioxide
Physical
- Colourless, odourless, tasteless
- Slightly soluble in water
- More dense (heavier) than air
- Sublimes Chemical
- Turns limewater milky
- Does not support combustion
- Acidic
- Dissolves in water to form carbonic acid (fizzy drinks)
The test for carbon dioxide is that it turns limewater milky
Word equation = Limewater + carbon dioxide ------> Calcium carbonate + water
Chemical equation = Ca(OH)2 +CO2 ------> CaCO3 +H2O
Uses of carbon dioxide
Chemical equation = Ca(OH)2 +CO2 ------> CaCO3 +H2O
Uses of carbon dioxide
- Stage effects - Dry ice
- Fire extinguishers
- Fizzy drinks
- Refrigeration (dry ice)
- Photosynthesis
Chapter 8- water
Water test-
Properties of water
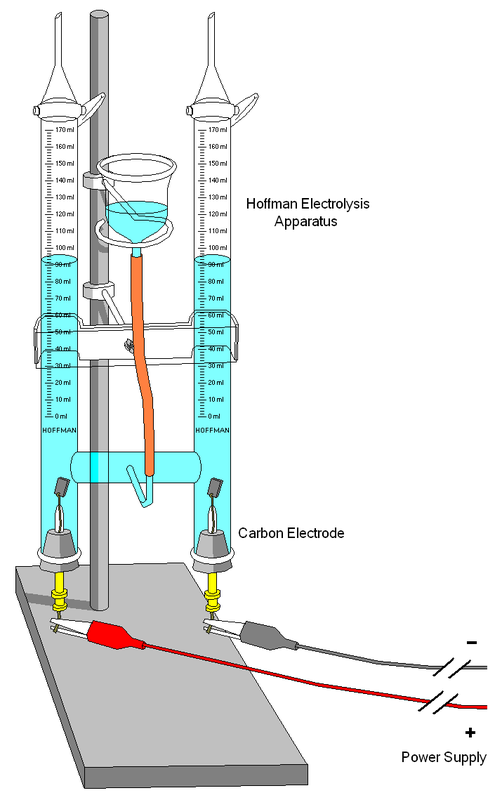
Electrolysis is the splitting up of a compound by passing electricity through it
- Turns blue cobalt chloride paper pink
Properties of water
- Water is made up of two elements -hydrogen and oxygen
- Turns cobalt chloride paper from blue to pink
- Turns anhydrous (no water) copper sulfate from white to blue
- Colourless, odorless
- A pH of 7
- Density = 1 g/cm 3 @ 4*C
- Excellent solvent
- Boiling point of 100*C
- Can be split using electrolysis
Electrolysis is the splitting up of a compound by passing electricity through it
Pure water is a poor electrical conductor so a little bit of sulfuric acid is added
After a while it will become clear that there is twice the amount of hydrogen than oxygen as as the water levels drop, one side will read half the amount of the other side
H2 : O2
2 : 1
Expansion of water - Usually substances expand when heated and contract when cooled
water behaves like this until 4*C however after this instead of contracting it expands
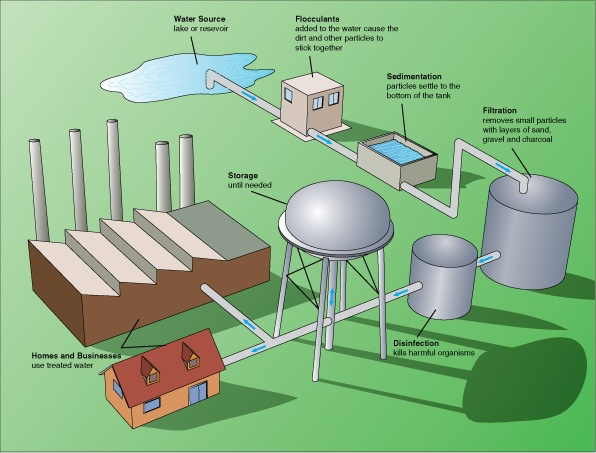
Treatment of water
After a while it will become clear that there is twice the amount of hydrogen than oxygen as as the water levels drop, one side will read half the amount of the other side
H2 : O2
2 : 1
Expansion of water - Usually substances expand when heated and contract when cooled
water behaves like this until 4*C however after this instead of contracting it expands
Treatment of water
[ N.B there is also a procedure called fluoridation not shown on the diagram in which fluoride is added to the water to help improve teeth and gum health (fluoride is in toothpaste) ]
UNIT 3
28. Atomic Structure
29. The Periodic Table
30. Chemical Bonding
31. Chemical Formulas and Chemical Equations
32. Metals and Plastics
28. Atomic Structure
29. The Periodic Table
30. Chemical Bonding
31. Chemical Formulas and Chemical Equations
32. Metals and Plastics
Chapter 9 - Atomic Structure
|
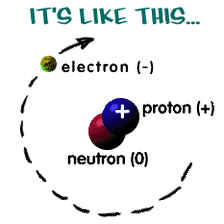
The Atom:The atom is the smallest unit that makes up the chemical elements. Every material object, or substance that can be touched and felt, is made up of atoms. Everything that is solid, liquid, or gas is made up of atoms.
An atom can only be seen through an Electron Microscope. Seen to the right are gold atoms through an electron microscope. |
|
Atomic Structure of the First 20 Elements
Number of Electrons:
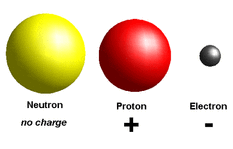
All atoms must be neutral, Therefore the number of (negative) Electrons have to be equal to the number of (positive) Protons.
No. Electrons = No. Protons
Number of Electrons:
All atoms must be neutral, Therefore the number of (negative) Electrons have to be equal to the number of (positive) Protons.
No. Electrons = No. Protons
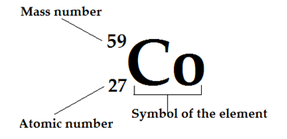
Nuclear Formula
The Atomic and mass numbers are often written with the symbol of an element.
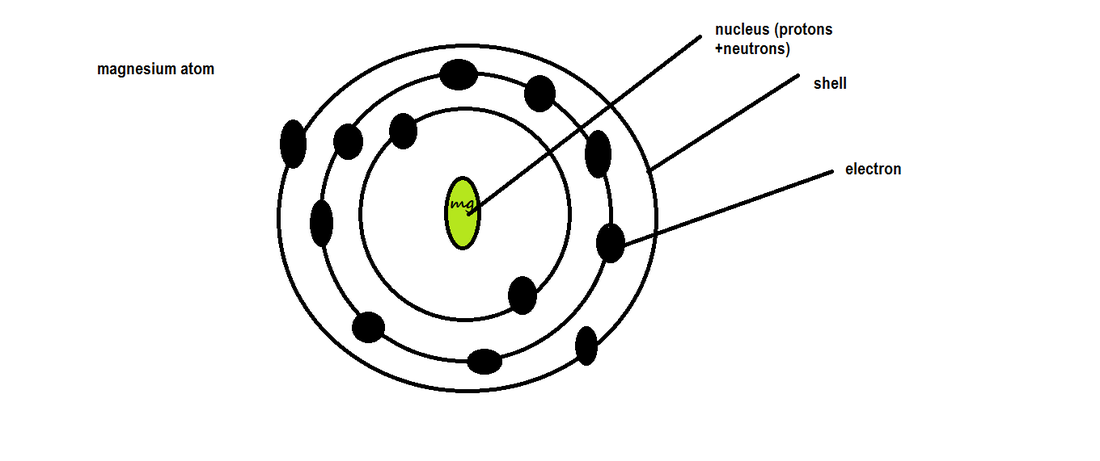
Bohr structure
When an atom is drawn , electrons are drawn as either 'Xs' or small circles (dots)
The 1st shell can only contain 2 electrons , the other shells can have upto 8 electrons
The Atomic and mass numbers are often written with the symbol of an element.
Bohr structure
When an atom is drawn , electrons are drawn as either 'Xs' or small circles (dots)
The 1st shell can only contain 2 electrons , the other shells can have upto 8 electrons
Isotopes
Isotopes are atoms of the same element that have the same atomic no. but different mass numbers
This is because of different numbers of neutrons in the nucleus
E.G Carbon 12 and Carbon 13
6 p 6 p
6 e 6 e
6 n 7n
Isotopes are atoms of the same element that have the same atomic no. but different mass numbers
This is because of different numbers of neutrons in the nucleus
E.G Carbon 12 and Carbon 13
6 p 6 p
6 e 6 e
6 n 7n
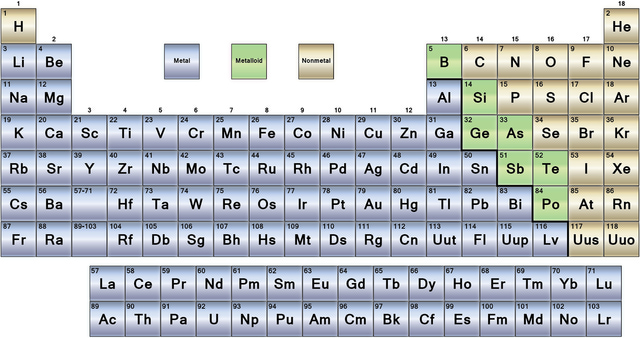
Chapter 10:The periodic table
Vertical lines = groups
Horizontal lines = periods
Alkali metals.
Reactions of alkali metals
Reactions with water...
Other important groups to know-
Horizontal lines = periods
Alkali metals.
- The alkali metals are group 1 metals.
- They have 1 electron in their outer shell
- soft metals - cut easily
- low melting points
- low densities - float
- shiny
- very reactive with air and water
- stored under oil
- form basic oxides
Reactions of alkali metals
Reactions with water...
- sodium + water------->sodium hydroxide + hydrogen
- sodium + oxygen-------->sodium oxide
Other important groups to know-
- Group 8 (the noble gases) are stable i.e they have a full outer shell of electrons
Chapter 11 - Chemical bonding

Atoms join together because of the octet rule which states that.....
all atoms want a full outer shell of electrons.
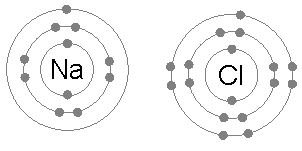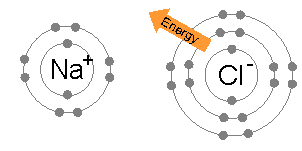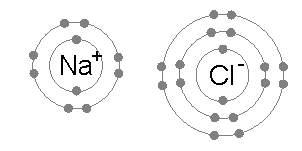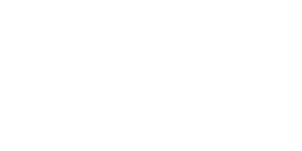
For example a sodium (Na) atom has the atomic structure 2 , 8 , 1 meaning it has 1 electron on its outer shell
because of the octet rule sodium joins with chlorine (Cl) to form sodium chloride (NaCl) A.K.A table salt
Bonding
bonding means the way in which atoms join together
there are 2 types of bonding , ionic and covalent
Ionic bonding
- ionic bonding is the transfer of electrons
- an atom that has lost or gained an electron is called an ion , and is charged
- This is how sodium and chlorine bond
- sodium loses an electron as it has one on its outer shell and chlorine gains an electron as it has 7 on its outer shell
- because sodium LOST an electron it is POSITIVELY charged (+)
- because chlorine GAINED an electron it is NEGATIVELY charged (-)
- They join together to form NaCL( sodium chloride)
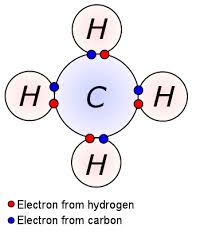
Covalent bonding
- covalent bonding is the sharing of electrons
- e.g Methane (CH4)
- in this example carbon shares the 4 electrons on its outer shell with 4 hydrogens to have a full outer shell
- each hydrogen atom needs 1 more electron to have a full outer shell so it shares it with one carbon electron
|
Properties of ionic and covalent compounds
Ionic
Covalent
Valency
|
|
Chapter 12 - chemical equations and chemical formulaes
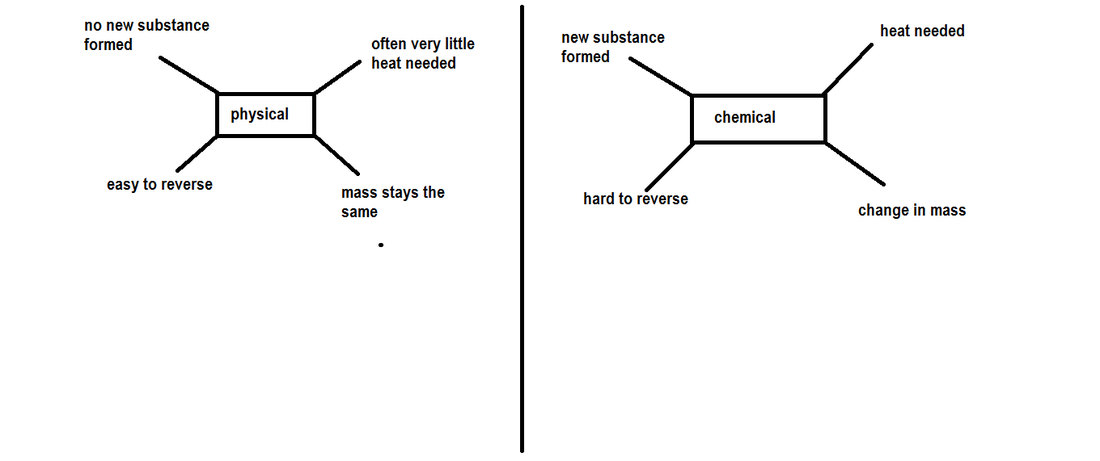
Every day things change chemically e.g water turning to ice
There are two types of changes
There are two types of changes
- Physical - where no new substance is formed
- E.G ice melting
- Chemical - a new substance is formed
- E.G burning coal in a fire
Chemical reactions -
Chemical formula
Word formula
Chemical equations are much quicker and easier to write so scientists mainly use this method of recording chemical equations
However , we must make sure these equations are balanced
Balancing equations
E.G Mg + O2 ------------>MgO
This equation is not balanced
There are 2 atoms of oxygen on the left but only 1 on the right. DO NOT write MgO2 as this is wrong instead write 2MgO
This means that there are 2 atoms of oxygen on the right but it also means 2 atoms of Mg so go back to the left and write 2Mg
The equation is now balanced correctly-
2Mg + O2 ---------->2MgO
- To record Chemical reactions we use 2 ways- chemical formula's and word formula's
Chemical formula
- HCL + NaOH ---------> NaCL + H2O
Word formula
- Hydrochloric acid + Sodium hydroxide -------------> Sodium chloride + Water
Chemical equations are much quicker and easier to write so scientists mainly use this method of recording chemical equations
However , we must make sure these equations are balanced
Balancing equations
- An equation is balanced when the total No. of atoms on the left hand side is equal to the total No. of atoms on the right hand side
E.G Mg + O2 ------------>MgO
This equation is not balanced
There are 2 atoms of oxygen on the left but only 1 on the right. DO NOT write MgO2 as this is wrong instead write 2MgO
This means that there are 2 atoms of oxygen on the right but it also means 2 atoms of Mg so go back to the left and write 2Mg
The equation is now balanced correctly-
2Mg + O2 ---------->2MgO
Chapter 13 - Metals and plastics
|
Properties of metals
Examples include > Iron , lithium , copper , aluminium , silver Alloys An alloy is a mixture of metals examples >
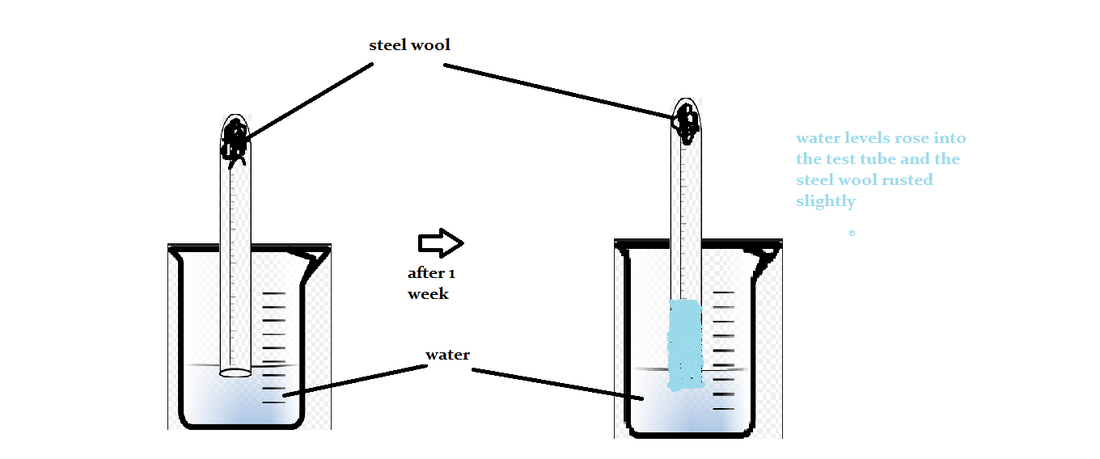
Corrosion and rusting
Reactivity of metals
-The Reactivity order is (1 being the most reactive)
Plastics Properties -
How do we make plastics
Everyday examples of Plastics are -----> Bin bags phone backs Impact of Plastics on the environment
|